Faster Answers for Rare Disease
WGS has been shown to have a superior diagnostic yield, (27%-59%)* compared to standard testing (12%-24%)*, potentially providing patients a higher likelihood of receiving a diagnosis.3-7, 15
With its improved diagnostic performance, WGS holds the promise of faster answers, helping patients and their families end an odyssey of inconclusive testing and focus on clinical care.
*diagnostic yield may vary based on cohort and individual disease states.
First-Line GWS vs. non-GWS
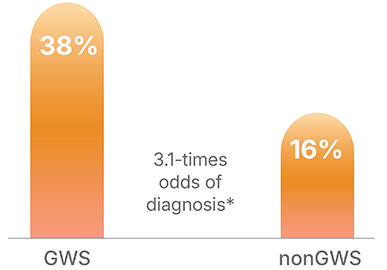
*within cohort comparisons in 10 studies; 95% CI: p<0.05)
GWS: Genome-wide sequencing; GS: Genome sequencing; ES: Exome sequencing
Based on meta-analysis of 108 studies comprising 24,631 children.15
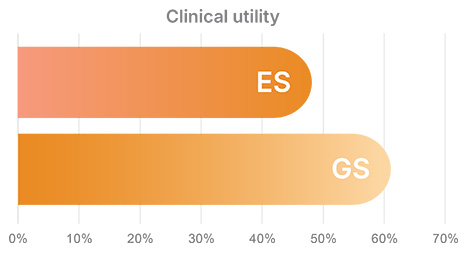
In a meta-analysis of literature from 2012-2021 featuring 159 cohorts and 50,417 probands, clinical utility was significantly greater following GS vs. ES*,15
*Overall pooled DY not significantly different between ES and GS
Improved Clinical Management
Standard of care (SOC) genetic testing such as chromosomal microarray or targeted panels are less likely than WGS to result in a diagnosis that would impact clinical management.9,13
WGS has been shown to impact clinical management in outpatient settings and when implemented early in the diagnostic pathway, has the potential to lead to life-changing options that care providers can offer to their patients.2 A change in management has been reported in 49–75% of pediatric outpatients who received a diagnosis by WGS.10-11

Knowledge for Diagnosis and Treatment is Growing
Approximately 250 new genes with disease-causing variation are added to the literature annually13 and WGS-enabled diagnoses can lead to a growing number of effective interventions in acute care settings.


One test to replace the many
An Early Diagnosis Can Prevent a Long Diagnostic Odyssey
With its improved diagnostic performance and faster time to answer, WGS holds the promise of helping patients and their families end an odyssey of inconclusive testing and unnecessary medical interventions—or prevent one altogether—and focus on care management.
References
- Palmer EE, Sachdev R, Macintosh R, et al. Diagnostic Yield of Whole Genome Sequencing After Nondiagnostic Exome Sequencing or Gene Panel in Developmental and Epileptic Encephalopathies. Neurology. 2021;96(13):e1770-e1782. doi:10.1212/ 0000000000011655
- Owen MJ, Niemi AK, Dimmock DP, et al. Rapid Sequencing-Based Diagnosis of Thiamine Metabolism Dysfunction Syndrome. N Engl J Med. 2021;384(22):2159-2161. doi:10.1056/NEJMc2100365
- Lindstrand A, Eisfeldt J, Pettersson M, et al. From cytogenetics to cytogenomics: whole-genome sequencing as a first-line test comprehensively captures the diverse spectrum of disease-causing genetic variation underlying intellectual disability. Genome Med. 2019;11(1):68. doi:10.1186/s13073-019-0675-1
- Vanderver A, Bernard G, Helman G, et al. Randomized Clinical Trial of First-Line Genome Sequencing in Pediatric White Matter Disorders. Ann Neurol. 2020;88(2):264-273. doi:10.1002/ana.25757
- Costain G, Jobling R, Walker S, et al. Periodic reanalysis of whole-genome sequencing data enhances the diagnostic advantage over standard clinical genetic testing. Eur J Hum Genet. 2018;26(5):740-744. doi:10.1038/s41431-018-0114-6
- Stavropoulos DJ, Merico D, Jobling R, et al. Whole Genome Sequencing Expands Diagnostic Utility and Improves ClinicalManagement in Pediatric Medicine. NPJ Genom Med. 2016;1:15012-. doi:10.1038/npjgenmed.2015.12
- Lionel AC, Costain G, Monfared N, et al. Improved diagnostic yield compared with targeted gene sequencing panels suggests a role for whole-genome sequencing as a first-tier genetic test. Genet Med. 2018;20(4):435-443. doi:10.1038/gim.2017.119
- Soden SE, Saunders CJ, Willig LK, et al. Effectiveness of exome and genome sequencing guided by acuity of illness for diagnosis of neurodevelopmental disorders. Sci Transl Med. 2014 Dec 3;6(265):265ra168. doi: 10.1126/scitranslmed.3010076. PMID: 25473036; PMCID: PMC4286868.
- Yeung A, Tan NB, Tan TY, et al. A cost-effectiveness analysis of genomic sequencing in a prospective versus historical cohort of complex pediatric patients. Genet Med. 2020 Dec;22(12):1986-1993. doi: 10.1038/s41436-020-0929-8. Epub 2020 Aug 10. PMID: 32773771.
- Scocchia A, Wigby KM, Masser-Frye D, et al. Clinical whole genome sequencing as a first-tier test at a resource-limited dysmorphology clinic in Mexico. NPJ Genom Med. 2019;4:5. Published 2019 Feb 14. doi:10.1038/s41525-018-0076-1
- Bick D, Fraser PC, Gutzeit MF, et al. Successful application of whole genome sequencing in a medical genetics clinic. J Pediatr Genet. 2016;6(2):61-76.
- Seaby EG, Rehm HL, O’Donnell-Luria A. Strategies to Uplift Novel Mendelian Gene Discovery for Improved Clinical Outcomes. Frontiers in Genetics. 2021;12. doi:10.3389/ fgene.2021.674295
- Clark MM, Stark Z, Farnaes L, et al. Meta-analysis of the diagnostic and clinical utility of genome and exome sequencing and chromosomal microarray in children with suspected genetic diseases. NPJ Genomic Med. 2018;3:16. doi. org/10.1038/s41525-018-0053-8
- Owen, M.J., Lefebvre, S., Hansen, C. et al. An automated 13.5-hour system for scalable diagnosis and acute management guidance for genetic diseases. Nat Commun 13, 4057 (2022). https://doi.org/10.1038/s41467-022-31446-6
- Pandey R et al. A meta-analysis of diagnostic yield and clinical utility of genome and exome sequencing in pediatric rare and undiagnosed genetic diseases, Genetics in Medicine (2025), doi: https://doi.org/10.1016/j.gim.2025.101398.

